New research highlights clinical importance of interaction between immune cells in the lymphoma microenvironment
The efficacy of immunotherapies in the most common lymphoid malignancy, diffuse large B-cell lymphoma, has been modest. New findings on how immune cells communicate could inform a more accurate way to group patients for treatment.
Approximately 700 people are diagnosed with diffuse large B-cell lymphoma (DLBCL) in Finland each year. It is the most common cancer that originates from lymphocytes, immune system cells. Although the disease is treated with the goal of achieving complete remission and preventing recurrence, a third of the patients experience progression and have a dismal prognosis. One of the challenges in treatment is that DLBCL is a heterogeneous disease, and better tools are needed for grouping the patients for adequate treatment.
Considering the success of immunotherapies in solid tumors and Hodgkin lymphoma, tumor-infiltrating immune cells are attractive therapeutic targets also in DLBCL. These checkpoint blockage therapiescan help the body’s immune system to recognize and attack cancer cells.
However, the efficacy of checkpoint blockage therapies against DLBCL has thus far been modest. This is mainly because the mechanisms of how immune cells and lymphoma cells communicate with each other have been unknown. Understanding this better would help to inform strategies for the use of immunotherapies.
Decoding the composition of tumor infiltrating immune cells
Now, researchers at the University of Helsinki and Helsinki University Hospital Comprehensive Cancer Centre have decoded the heterogeneity of tumor infiltrating immune cell composition in DLBCL.
In addition to genomic alterations in the tumor cells, the development of the disease is impacted by the tissues surrounding cancer cells, the lymphoma microenvironment. It consists of blood vessels, extracellular stroma, and immune cells, including T lymphocytes and macrophages.
The study, recently published in Clinical Cancer Research, investigated the interplay between these immune cells in the lymphoma microenvironment. By utilizing novel imaging technologies, gene expression profiling and advanced data analyses, the researchers characterized lymphoma infiltrating T cells and macrophages, and their spatial interactions in 178 lymphoma samples at the single cell level. The findings were correlated with patient demographics and survival. The results were validated in two external cohorts comprising a total of 633 patients.
Clinically meaningful interplay between immune cells
Researchers found that based on the immune cell content, DLBCL can be divided into inflamed and noninflamed subtypes. These are characterized by high and low immune cell contents, respectively. DLBCL patients with a high T cell content but a low macrophage content had favorable survival. However, when macrophages expressed immune checkpoint molecules, inflamed subtype translated to inferior outcome.
They also discovered that certain checkpoint molecules are enriched on macrophages when they interact with T cells, suggesting that checkpoint molecules are central in the interplay between macrophages and T cells.
The findings extend previous studies, which have mostly focused on characterizing the lymphoma microenvironment based on gene expression. The results demonstrate that the interplay between immune cells in the lymphoma microenvironment is clinically meaningful.
In particular, the results highlight the adverse impact of checkpoint molecule expressing macrophages on the antitumoral activity of T cells. The findings also suggest that checkpoint blockage therapy should be investigated in clinical trials in patients with inflamed DLBCLs and high proportions of checkpoint molecule expressing macrophages. In the longer term, this could inform a more accurate way to group DLBCL patients for immunotherapies.
Original publication:
Matias Autio, Suvi-Katri Leivonen, Oscar Brück, Marja-Liisa Karjalainen-Lindsberg, Teijo Pellinen, and Sirpa Leppä. Clinical impact of immune cells and their spatial interactions in diffuse large B-cell lymphoma microenvironment. Clinical Cancer Research, 2022.
DOI: 10.1158/1078-0432.CCR-21-3140
The study was primarily funded by the University of Helsinki, Helsinki University Hospital, Academy of Finland, iCAN Digital Precision Cancer Medicine Flagship, Sigrid Jusélius Foundation and the Finnish Cancer Organizations.
Further information:
Sirpa Leppä, Professor, Dept of Oncology, University of Helsinki, and Helsinki University Hospital Comprehensive Cancer Centre
sirpa.leppa@helsinki.fi
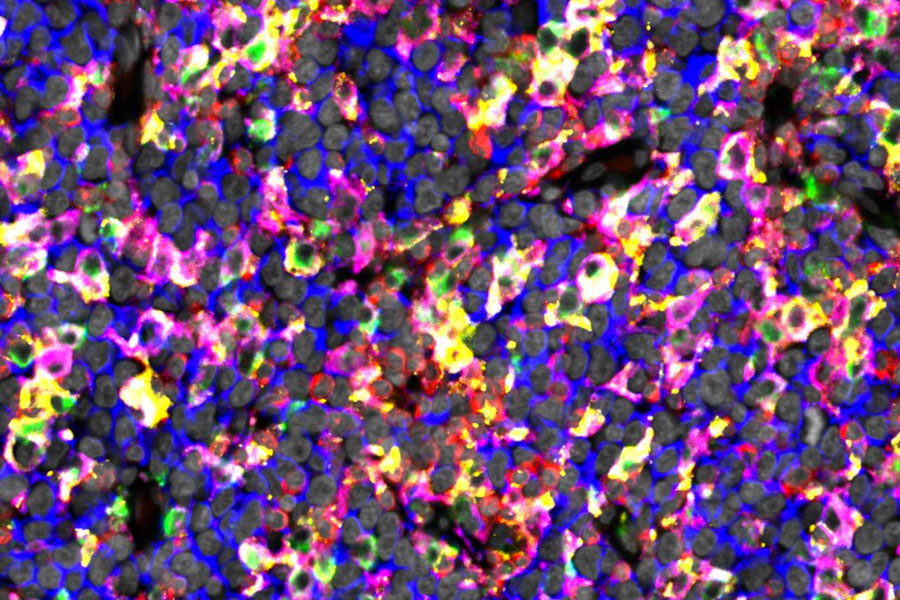
The figure shows immunostaining of lymphoma tissue rich in checkpoint proteins expressing macrophages. The image depicts CD20 positive lymphoma cells in blue, macrophage markers CD68 and CD163 in green and in magenta, checkpoint molecules TIM3 and PD-L1 in red and yellow, and DAPI nuclear stain in gray.